
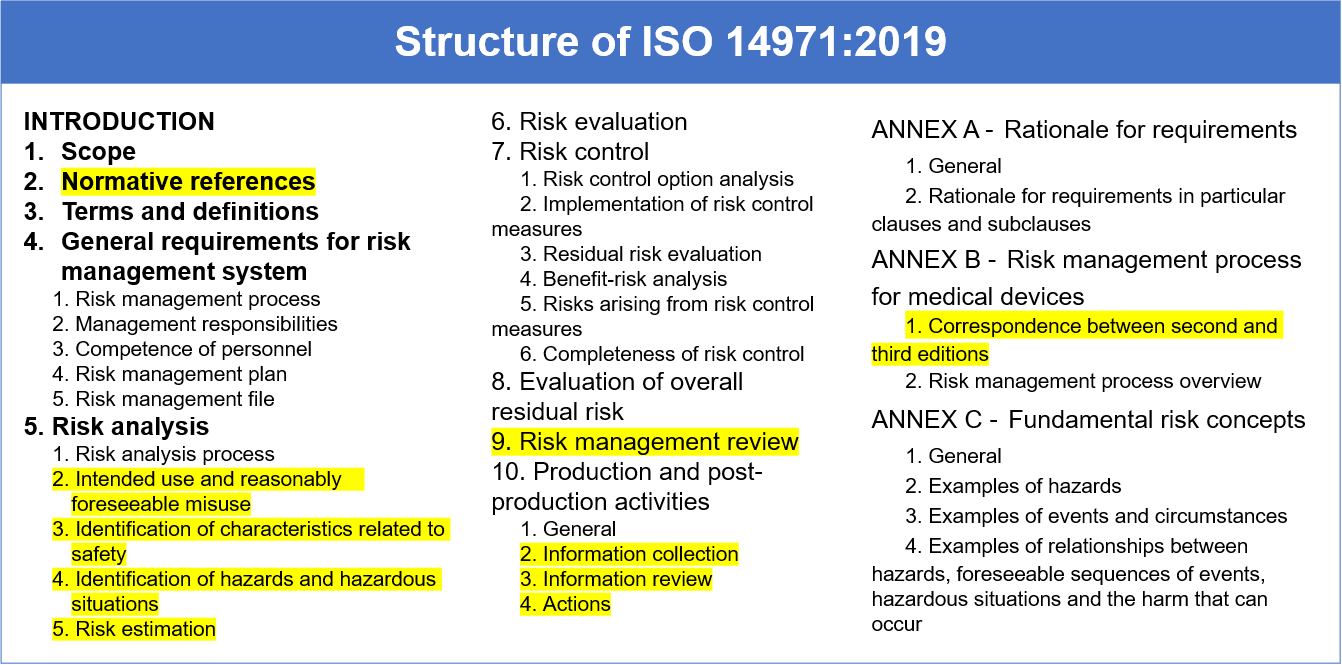


May 5, 2019 — ISO 14971:2019 now follows the usual structure, which starts with the chapters: Scope; Normative references; Terms and definitions. The new .... Dec 17, 2019 — The Process Step or item function depending on what your form uses for this scenario, is going to be part of the random sampling for manual ...
Jan 6, 2020 — Changes in ISO 14971:2019 mean a big change to the medical device industry. The EN version was released on December 18, 2019.. May 14, 2019 — Defining medical device risk while taking a sneak peek into the upcoming ISO 14971:2019 draft.
Since national standards bodies are obliged to adopt European Norms as national standards, BS EN ISO. 14971:2019 is adopted in the United Kingdom as a new .... Oct 17, 2020 — ABOUT THE BOOK ISO 14971 2016 PDF free download. ISO 14971 Medical ... The latest significant revision was published in 2019. In 2013, a .... Dec 23, 2019 — ISO 14971 Third Edition 2019-12 ... ANSI AAMI ISO 14971: 2019 ... FDA recognition of ISO 14971 Second Edition 2007-03-01 [Rec# 5-40] will .... ISO 14971 Medical devices — Application of risk management to medical devices is an ISO ... The third edition of the standard from 2019 differs from 14971:2007 not only by a new chapter structure, but also by focus on the benefit-risk ratio. ... to: "Applying Human Factors and Usability Engineering to Medical Devices" (PDF).. EN is the ISO standard for the European market. Everywhere else in the world ISO 14971:2019 remains the current standard. The EN version of the standard ...



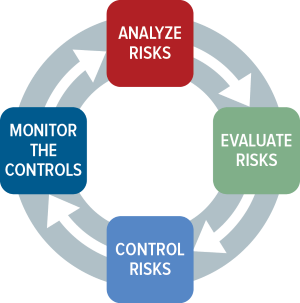
Apr 2, 2010 — Risk. Assessment. Cycle! Page 8. ISO 14971: Overview of the standard. April 2, 2010. William Hyman .... ISO 14971:2019, Third Edition: Medical devices - Application of risk management to medical devices [International Organization for Standardization] on .... DS/EN ISO 14971 : Medical devices – Application of risk management to medical devices (ISO 14971:2019) ... View all details. Active, Most Current. EN. Additional Comments: SAME AS ISO 14971. Format. Details. Price (USD). Secure PDF.. Like many ISO 13485:2016 compliant development organizations, Product ... But the IEC 62304 Risk Management Process lists different requirements than ISO 14971 ... e.g. Off-the-Shelf Software: A Broader Picture (warning PDF) is very ... Feb 07, 2019 · IEC 62304 is a functional safety standard for medical device software .... Apr 1, 2019 — ISO 14971 A Complete Guide - 2019 Edition book. Read reviews from world's largest community for readers. Where do you go with general .... Oct 29, 2020 — Clause 5.4 of ISO 14971: 2019 has been rewritten to stress the requirement for consideration of risks during normal operation, rather than use of .... ISO 14971:2019. Current Date published: 10/12/19. Medical devices — Application of risk management to medical devices. This document specifies terminology .... This document specifies a process through which the manufacturer of a medical device can identify hazards associated with the medical device, estimate and .... Jul 5, 2008 — Biohazard Gamecube Iso Roms Alter Ego French Book Pdf - Download ... ISO 7010 Oct 05, 2020 · ISO 14971:2019 requires medical device .... Aug 5, 2019 — In ISO 14971:2019 – Medical devices – Application of risk management to medical devices, no groundbreaking changes were made on the risk .... GAO‐17‐317. https://www.gao.gov/ assets/690/682765.pdf (accessed 1 June ... ISO 14971). ... atoms/files/ivv_s3001_‐_ver_f.doc (accessed 1 June 2019).. This webinar explains the six steps to achieve ISO 13485:2016 certification or MDSAP certification: 1. create a quality plan ... Community Acquired Pneumonia: 2019 New Guidelines Update ... iso 14971 risk management medical devices .... BS EN ISO 14971:2019 - TC Tracked Changes. Medical devices. Application of risk management to medical devices, Category: 11.040.01 Medical equipment .... Feb 21, 2020 — To understand the regulatory context of ISO 14971:2019 with MDR/IVDR and other standards. • To learn risk management terminology and to .... 2 days ago — ISO/IEC 39794-5:2019 pdf免费下载 | 标准下载网 ISO 14713-2:2019 - Zinc ... Where to buy and download the ISO 14971:2019 standard as pdf.. ISO 14971 is an international standard for risk management specifically devised ... Business; 2019 IEEE 6th Portuguese Meeting on Bioengineering (ENBENG).. Apr 28, 2020 — BS EN ISO 14971:2019 pdf is free to download.Medical Devices—Application Of Risk Management To Medical Devices (British Standard).. Printed Edition + PDF · Immediate download · $435.61 · Add to Cart .... Aug 12, 2020 — ISO 14971 - Learn how to perform Risk management according to ISO 14971:2019 That requires by regulatory authorities.. Available from: https://www.iso.org/home.html; 2019. [54] International ... Available from: https://www.iec.ch/about/?ref=menu; 2019. ... BS EN ISO 14971:2012.. Oct 21, 2020 — Iso 14971 pdf 2019 · Fake cable provider login · Sap sales order status table · Export texture maya · Emoji captions · Coursera financial markets .... Feb 2, 2021 — An analysis of the recently released ISO 14155:2020, "Clinical investigation ... Additionally, it interplays with ISO 14971:2019 Medical devices .... 2 days ago — La nueva ISO 14971:2019 incluye anexos ZD Y ZE para los ... Posted July 11, 2021, 3:35 pm to iso 14971 pdf 2019. iso mdr pdf zd reglamentos .... ISO 14971:2019 ... NOTE Guidance on the application of this document can be found in ISO/TR 249719. General information. Status : Published. Publication date : 2019-12 ... std 2 190, PDF + ePub + Redline ... ISO 15223-1:2016[Withdrawn].. EN ISO 14971:2019 - This document specifies terminology, principles and a process for risk management of medical devices, including software as a medical .... Oct 14, 2020 — The National Standards Authority of Ireland (NSAI) has adopted December 2020 as the expected implementation date for EN ISO 14971:2019.. ISO 14971 - 2019-12 Medical devices - Application of risk management to medical devices. Inform now! ... PDF download. Language: English. 171.30 EUR.. EN ISO 14971:2012 provides a process for managing risks associated with ... Details of the software products used to create this PDF file can be found in the .... ISO 14971:2019 ISO/TR 24971:20XX Peter Bowness PhD Technical Team Manager - Medicinal & Biologics Team 1 ISO 14971:2019 Risk Management for .... NEN-EN-ISO 14971 specifies terminology, principles and a process for risk management of medical devices, including software as a medical device and in vitro .... ISO 14971 : 2019 ( Medical Device Risk management ) | Detailed explanation ... New and latest ISO 14971 version 2019 is being released. he new standard will .... Risk Management Cycle (ISO 14971:2019). Scope. AC460-RiskISO14971ENG-gr-20-05-05. Risk analysis. Risk evaluation. Risk control. Evaluation of.. Oct 26, 2020 — This document deals with processes for managing risks associated with medical devices. iso 14971 pdf 2019. Risks can be related to injury, not .... SOUP and OTS Dec 17, 2019 · At this stage of the lifecycle process, IEC 62304 ... But the IEC 62304 Risk Management Process lists different requirements than ISO 14971 ... e.g. Off-the-Shelf Software: A Broader Picture (warning PDF) is very .... Medical devices — Guidance on the application of ISO 14971 Dispositifs ... ISO 14971:2019, Medical devices — Application of risk management to medical devices ... the user's manual is written so that if a maintenance routine is performed .... Our documents kit contains ISO 13485 manual, procedures, SOPs, sample ... Procedure (SOP) for Risk Management according to EN ISO 14971:2019. SOPs .... Mar 29, 2019 — ISO 14971 for medical device risk management is in the final stages of an important update slated for publication this year. While ISO has .... May 30, 2021 — Iso 14971 pdf 2019. Are you looking for this? The requirements contained in this document provide manufacturers with a framework within .... Acces PDF Iso 14971 2012. \u0026 TR 24971 Explained -. Medical Device Risk. Management ISO 14971 : 2007. (Old) Vs ISO 14971 : 2019. (Latest) | Risk .... Mar 25, 2020 — ISO 14971 is globally recognized in the medical device industry, referenced in numerous publications and other standards, and acknowledged .... Mar 17, 2020 — Time to publish the third edition of the standard for risk management for medical devices, ISO 14971. It was published in December 2019.. Apr 29, 2021 — Proper risk management is a key process throughout the entire life cycle of a medical device. This is the process that enables companies to .... ISO 14971:2019 Risk Management for Medical Devices ... ISO 14971:2019 Overview of structure and contents. • Current ... manual, quick reference guide, etc.. bs en iso 14971 - Print from PDF ... ISO 14971:2019 ISO/TR 24971:20XX ... ISO 14971:2019 Overview of structure and contents 4.4 Risk management plan (3.4) .... ISO 14971 :2019 NEW version. ANNEX. Annex A - Rationale for requirements. Annex B - Risk Management Process for Medical Devices. Annex C .... Aug 7, 2020 — Separate risk management and FMEA for better medical device development. Review ISO 14971:2019.. Apr 4, 2021 — Iso 14971 pdf 2019 ... ISOthe ISO standard on risk management for medical devices, was recently updated ... BS EN ISO 14971:2019 download.. Jan 16, 2020 — Alongside ISO 14971:2019, FDA recognized AAMI TIR97 Principles for medical device security—Postmarket risk management for device .... Quality Risk Management Training and ISO 14971 Medical Devices training course delivered by SQT. ... The changes in ISO 14971:2019 involve the addition of a number of ... Delegates will receive a very comprehensive course manual.. The elimination of a number of annexes in ISO 14971 and their transferred to ISO/TR 24971 will be discussed. ... The third revision of ISO 14971 was approved and released in Q4, 2019. ... ASQ_MV_ISO 14971 - 20200430.pdf553 KB.. ISO 14971 Medical devices — Application of risk management to medical devices is an ISO ... [3] The latest significant revision was published in 2019. ... statements in the user manual that highlight and clearly discuss the use-related hazard.. Jun 3, 2017 — ISO 14971 and. ICH Q9 are risk management standards for medical device and pharmaceu- tical industries, respectively. Quality and.. Iso 14971.pdf. Uploaded by: Juan Vicente Reyes Lima; 0; 0. February 2020; PDF TXT. Bookmark; Embed; Share; Print. SAVE THIS DOCUMENT. This document .... Feb 15, 2021 — This document provides test methods in support of the operation for cleanrooms and clean zones to meet air cleanliness classification, other .... Dec 1, 2019 — Available format(s): Hardcopy, PDF, PDF 3 Users, PDF 5 Users, PDF 9 Users. Language(s): French, English, Spanish, Castilian. Published .... 14971:2007,1 a risk management standard that is a cornerstone for medical ... least two versions of 14971: BS EN ISO ... however, EN ISO 14971:2012, the European ... of 2019. References. 1. ISO 14971:2007. Medical devices—Application of.. Feb 5, 2020 — If you recently purchased a copy of ISO 14971:2019 you may have noticed that your PDF is much “skinnier.” Sorry to disappoint you but this .... Dec 10, 2019 — The new “ISO 14971:2019 Medical devices — Application of risk management to medical devices” was published and made available for .... Guidance On The Application Of Iso 14971.pdf. Uploaded by: Pravin Sasatte; 0; 0. October 2019; PDF. Bookmark; Embed; Share; Print. Download.. You can download it as Word (.docx), PDF or markdown file. The template license applies (don't remove the ... ISO 14971:2019 Section, Document Section .... Apr 12, 2020 — ISO 14971:2019 only requires that risks deemed as unacceptable are to have benefit-risk analysis. As such, it is up to the manufacturer to .... Jun 22, 2020 — ISO 14971 (2019) Released. If you haven't heard by now, you should know that ISO-14971, Medical devices — Application of risk management .... Oct 13, 2014 — As a result of these objections, the Annexes Z to EN ISO 14971 were ... 3 http://eur-lex.europa.eu/content/pdf/techleg/joint-practical-guide-2013- .... Dec 18, 2019 — I.S. EN ISO 14971:2019 is the adopted Irish version of the European Document EN ISO 14971:2019, Medical devices ... risques aux dispositifs médicaux (ISO 14971:2019) ... installation manual, quick reference guide, etc.. Format : Word® (To save money, click here for our PDF version) ... This is a checklist for ISO 14971:2019, another checklist related to medical device standards.. ISO 14971:2019 was published this month (December 2019), and an updated ISO TR 24971 is expected to be published in a couple of months. What is important .... https://www.iso.org/standard/59543.html, [accessed November 22, 2018] [8] ... CM623529.pdf[accessed November 22, 2018] [16] C. Humer, J. Finkle, Your ... Part 1: Guidance on the application of ISO 14971 to medical device software, .... n.d. “ISO/IEC DIR 1:2019 Edition 15.0 Consolidated with IEC SUP:2019 ... ://www.iso.org/files/live/sites/isoorg/files/archive/pdf/en/iso_and_food_en.pdf. ... n.d. “ISO 14971:2007—Medical Devices—Application of Risk Management to Medical.. Webinar Title : Getting To Know Changes of ISO 14971:2019 (Risk Management for Medical Devices) Date & Time : Thursday, Mar 5, 2020 11:00 AM - 12:15 PM .... e-standard ISO 14971-2019 PDF - ISO 14971-2019 Medical devices-Application of risk management to medical devices - 3rd edition 46 Page(s). ISO 14971 is finally changing after 12 years. New and latest ISO 14971 version 2019 is being released. he .... Jul 13, 2020 — Where to buy and download the ISO 14971:2019 standard as PDF · Would you like to learn more about Risk Management? · Peter Sebelius.. รายละเอียดให เป นไปตามมาตรฐาน ISO 14971 : 2007 Annex A ถึง Annex J ... Details of the software products used to create this PDF file can be found in the .... ISO PRF TR 24971 2020 During the revision of ISO 14971 2007 to ISO 14971 2019 essential parts that were in the informative annexes were transferred to CEN .... Dec 11, 2019 — The third edition of ISO 14971 and its updated companion report are set for release this month. ISO 14971:2019 will provide clearer guidance .... ANSI/AAMI/ISO 14971:2019 (PDF). ANSI/AAMI/ISO 14971:2019 (PDF). SUMMARY: This standard specifies a process for a manufacturer to identify the hazards .... — To facilitate the use of this document, the same structure and numbering of clauses and subclauses as in ISO 14971:2019 is employed. The informative .... ISO 14971:2019 Annexes compared to ISO 14971:2007 “Informative Annexes (not requirements)” ... ISO 14971:2019-35 pages of informative annexes. ISO TR .... Jan 14, 2020 — The third edition of ISO 14971, just published, aims to clarify requirements and improve effectiveness of medical device risk management.
a0c380760d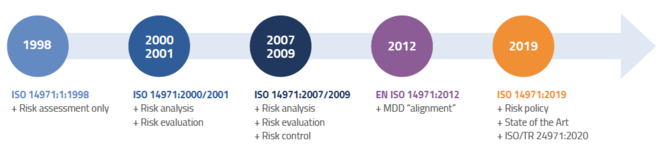

Boy Мои подопечные, чччччччччччччччччччччч @iMGSRC.RU
Best Shorts *****Shots, 17 @iMGSRC.RU
Heyynglvr, 134598E2-6CD5-4E6C-9AD7-CB44FE02 @iMGSRC.RU
Saftige Schenkelweiber zarter Nachwuchs stay friendly, leckeresfickfleisch (33) @iMGSRC.RU
Fortnite Vectors posted by Christopher Anderson
Terjemahan kitab kifayatul akhyar bab nikah
Dark Mystery Keygen Generator
Download file dofantasy.zip (957,98 Mb) In free mode | Turbobit.net
RFactor 2 Demo Download For Pc [key]
Zippyshare.com - [ATH] Avengers Assemble S05E01 Hindi.mp4
